|
2/28/2024 0 Comments Sigma bond
This is contrasted by sigma bonds which form bonding orbitals directly between the nuclei of the bonding atoms, resulting in greater overlap and a strong sigma bond. From the perspective of quantum mechanics, this bond's weakness is explained by significantly less overlap between the component p-orbitals due to their parallel orientation. The C-C double bond, composed of one sigma and one pi bond, has a bond energy less than twice that of a C-C single bond, indicating that the stability added by the pi bond is less than the stability of a sigma bond. Pi bonds are usually weaker than sigma bonds. Properties Two p-orbitals forming a π-bond. This latter mode forms part of the basis for metal-metal multiple bonding. One common form of this sort of bonding involves p orbitals themselves, though d orbitals also engage in pi bonding. 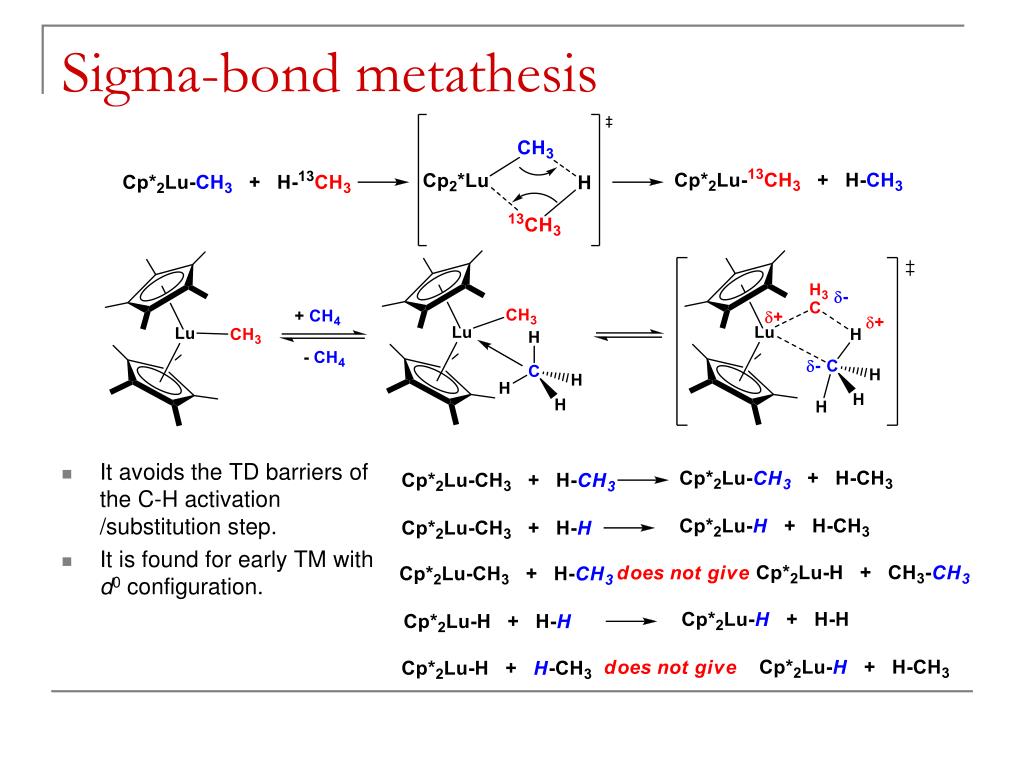
The Greek letter π in their name refers to p orbitals, since the orbital symmetry of the pi bond is the same as that of the p orbital when seen down the bond axis. Pi bonds can form in double and triple bonds but do not form in single bonds in most cases. 
This plane also is a nodal plane for the molecular orbital of the pi bond. Each of these atomic orbitals has an electron density of zero at a shared nodal plane that passes through the two bonded nuclei. In chemistry, pi bonds ( π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
